Effect of Graft Height Mismatch on Contact Pressures With Osteochondral Grafting of the Talus

L. Daniel Latt,y MD, PhD, Richard R. Glisson,z BS, Harvey E. Montijo,z MD, Federico G. Usuelli,§ MD, and Mark E. Easley,z MD
Investigation performed at the “Orthopaedic Research Laboratory”
Duke University, Durham, North Carolina
Background: Osteochondral allograft transplantation is technically demanding. It is not always possible to place the surface of the graft perfectly flush with the surrounding cartilage. One must often choose between placing at least some portion of the sur- face of the graft slightly elevated or recessed. The effect of this choice on joint contact pressure is unknown.
Purpose: This study was undertaken to determine the effect of graft height mismatch on joint contact pressure in the ankle.
Study Design: Controlled laboratory study.
Methods: Ten human cadaveric ankles underwent osteochondral grafting by removal then replacement of an osteochondral plug. Six conditions were tested: intact, graft flush, graft elevated 1.0 mm, graft elevated 0.5 mm, graft recessed 0.5 mm, and graft recessed 1.0 mm. Joint contact pressures were measured with a Tekscan sensor while loads of 200 N, 400 N, 600 N, and 800 N were sequentially applied.
Results: The peak contact pressure at the graft site for the flush condition was not significantly different from the intact condition for either medial or lateral lesions. In contrast, peak pressure on the opposite facet of the talar dome was significantly increased during the flush condition for the medial but not the lateral grafts. Elevated grafts experienced significantly increased contact pressures, whereas recessed grafts experienced significantly decreased pressures. These changes were greater for lateral than for medial lesions. Reciprocal changes in joint contact pressures were found on the opposite facet of the talus with elevated grafts on the lateral side and recessed grafts on the medial side.
Conclusion: Flush graft placement can restore near-normal joint contact pressure. Elevated graft placement leads to significant increases in joint contact pressure at the graft site. Recessed graft placement leads to a transfer of pressure from the graft site to the opposite facet of the talus.
Clinical Relevance: Osteochondral grafts in the talus should be placed flush if possible or else slightly recessed.
Keywords: osteochondral graft; talus; joint contact pressure; graft height mismatch
Osteochondral allograft transfer (OAT) is used to treat osteochondral lesions of the talus (OLTs) that have failed to respond to arthroscopic debridement combined with dril- ling or microfracture.3 The OAT procedure consists of cre- ating a cylindrical defect at the site of the OLT (the recipient site), harvesting an osteochondral graft from a nonweightbearing portion of the femoral condyles or talar dome, and then impacting the graft into the recipient site. This procedure has been shown to have 90% to 97% good-to-excellent medium- to long-term results in a number of studies.1,5,9,10,12 It is a technically demanding procedure. One of the most significant challenges is ensuring that the articular surface of the graft is made flush with the sur- rounding cartilage. One biomechanical study performed on porcine knees found that small differences in osteochon- dral graft height, particularly with an elevated graft, lead to significant increases in joint contact pressure at the graft site.7 A subsequent study by the same group exam- ined the effect of angled grafts and again found that eleva- tion led to increased pressures at the graft site.6 This information may be applicable to the human ankle joint; however, the shape of the porcine knee differs markedly from that of the human ankle, with the ankle cartilage experiencing a significantly greater load per unit of surface
area.11 Thus, it is anticipated that effects of articular sur- face incongruities in the ankle would be much greater than those found in the porcine knee. Moreover, these previous studies looked only at pressure at the graft site and not at the pressure distribution within the entire joint. The purpose of this study was to investigate the rela- tionship between talar graft height and peak contact pres- sures in the human ankle, both in the region of the osteochondral graft and elsewhere in the ankle joint. We hypothesized that placement of the articular surface of the graft either above or below the adjacent chondral sur- face would lead to an increase in joint contact pressure. We also hypothesized that changes in joint contact pressures on the grafted facet would be accompanied by reciprocal changes in pressure on the opposite facet of the talus.
Materials and methods
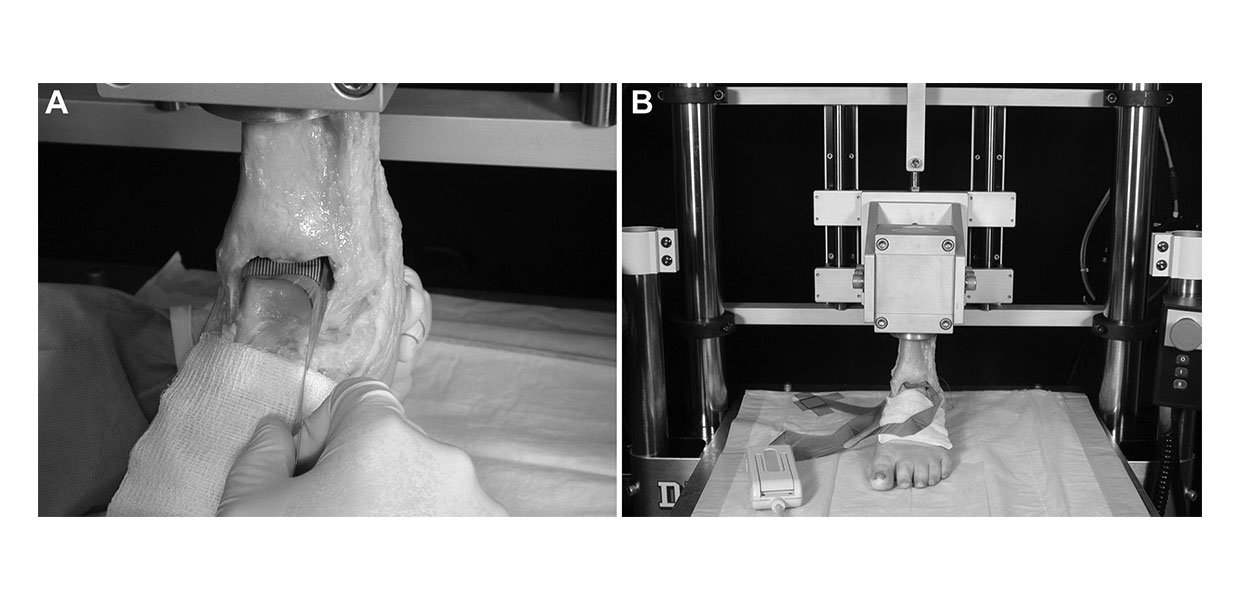
Ten fresh-frozen cadaveric feet were used. The donor age range was 33 to 70 years, with equal numbers of male and female donors. After thawing, the anterior soft tissues including the skin, subcutaneous tissue, tendons, neuro- vascular bundle, and anterior joint capsule were removed. Care was taken to ensure that the syndesmotic, deltoid, anterior talofibular, and calcaneofibular ligaments were left intact. The joint surface was inspected to ensure that it was free of chondral lesions. The proximal tibia and fib- ula were cleaned of soft tissue and then potted in an alumi- num tube using polyester auto body filler. Each specimen was mounted in a servohydraulic mate- rials testing machine (Model 1321, Instron Corp, Norwood, Massachusetts) for axial loading. The foot was placed in a neutral, plantigrade position on the testing machine table, with the axis of the second metatarsal oriented parallel to the sagittal plane through the foot (Figure 1B). The testing sequence consisted of applying axial loads from 0 to 800 N in increments of 200 N over a period of 55 seconds. Each incremental increase in load was maintained for 5 seconds before ramping up to the next load.
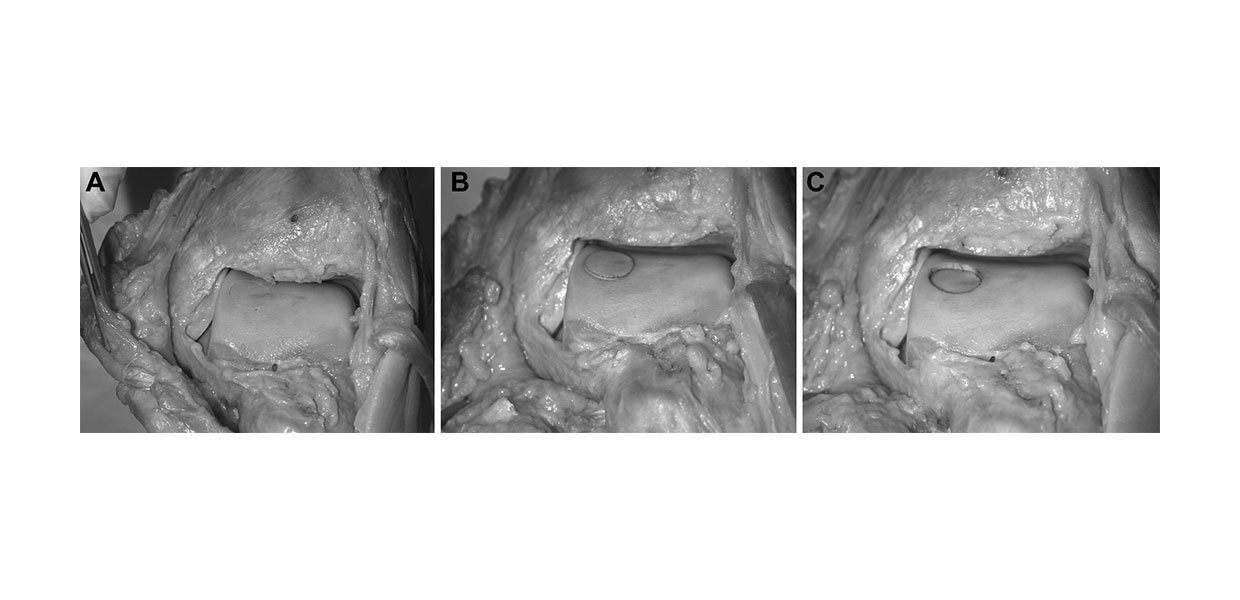
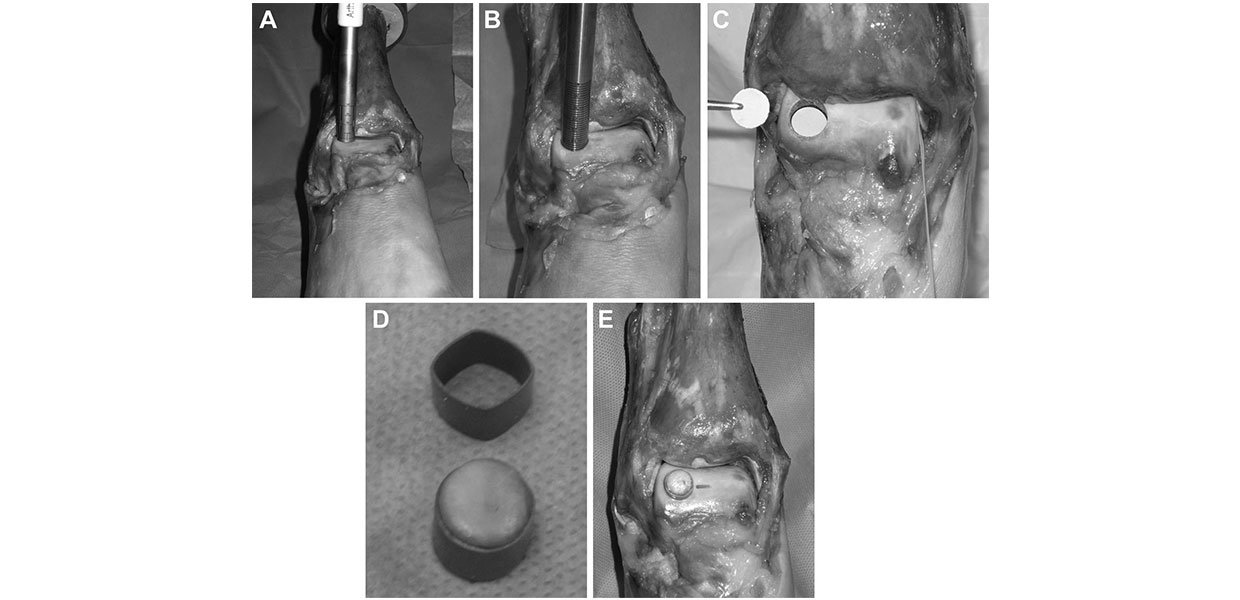
Each specimen was tested under 6 conditions, in the fol- lowing order: intact, graft flush, graft recessed 1.0 mm, graft recessed 0.5 mm, graft elevated 0.5 mm, and graft elevated 1.0 mm (Figure 2). The intact condition testing was performed before any modification of the joint surface. A medial (n = 5) or lateral (n = 5) defect was then created in the anteroposterior central third of the talar dome. These 2 locations were chosen because together they represent the site of approximately 80% of OLTs.4 At the selected graft site, the 10-mm recipient chisel from the osteochondral autograft transfer system (OATS, Arthrex, Inc, Naples, Florida) was used to remove a cylindrical osteochondral plug 10 to 12 mm in depth (Figure 3A). This excised osteo- chondral plug was subsequently used to fill the defect that had just been created, effectively eliminating as a variable the inevitable mismatched surface contours that would have resulted had the graft been obtained from a different site. Before harvest of the graft, its rotational orientation was marked with indelible ink (Figure 2B).
The osteochondral graft was placed in a custom-made sanding jig, and its base was sanded smooth and square to the sides of the cylinder on 240-grit wet-or-dry sandpa- per lubricated with saline. The base of the recess created in the talus by removal of the graft was similarly made flat and square to the sides using a cylindrical reamer and 10-mm OATS cylindrical tamp. Precision 10-mm– diameter circular aluminum shims of various thick- nesses were placed in the bottom of the recess so that the articular surface of the graft was perfectly flush with the surrounding articular surface when reinserted into the recess (Figure 3C). To prevent rocking of the graft within the recess, a polyvinyl chloride (PVC) sleeve with a wall thickness equaling the thickness of the chisel kerf was placed over the graft before placement in the recipient site (Figure 3D). The graft with its sleeve was then replaced into the defect (Figure 3E), the original rotational position was reestablished, and the graft was lightly impacted. After obtaining joint pressure maps in the flush graft condition, the recessed and elevated graft conditions were achieved by removal or addition, respectively, of 0.5-mm–thick shims at the base of the graft (Figure 2). The height of the step created by the shimming was verified by sweeping the region of the graft with a dial indicator. A Tekscan sensor (Knee Sensor #4000, Tekscan Inc, Boston, Massachusetts) interposed between the tibial and talar articular surfaces was used to collect real-time joint contact pressure maps (Figure 1A).
This 0.1-mm–thick sensor has a 10.34-MPa pressure range, 62 sensing ele- ments cm2, and a 2.8 3 3.3-cm rectangular area. Before use, each sensor was equilibrated and then calibrated with known pressures with a pneumatic calibrator. The pressure maps were recorded at 4 frames per second. The pressure maps obtained under each graft condition and joint load magnitude were analyzed using Tekscan iSCAN version 5.0 software. At each applied load, the peak contact pressure was determined in the region of the graft and on the opposite facet of the talar dome. A square 8 3 8 sensing element (approximately 10 3 10 mm) region of interest (ROI) was used to separate the pres- sure measurements at the graft site from the surrounding area. The location of the ROI at the graft site was deter- mined by visual inspection of the pressure maps. This loca- tion was usually readily apparent on the elevated and recessed trials. This position was then transferred to the corresponding intact trial. The location of the ROI on the opposite facet was centered over the region of greatest pressure on the opposite facet. The total force measured across the talar dome was calculated by summing the prod- uct of the pressure and area at each sensing element. The peak pressure measurements were normalized by multi- plying them by the ratio of the known load applied by the testing machine (200, 400, 600, and 800 N) to the force calculated from the sensor output, thus negating any non- linearity or drift in sensor calibration. The peak pressure difference between the intact condition and the experimental graft conditions was then calculated as the normalized peak pressure from each graft condition less the normal- ized peak pressure in that location from the intact trial. A peak pressure ratio (expressed as a percentage) was also calculated, using the normalized peak pressure from each graft condition divided by the corresponding value from the intact condition multiplied by 100.
Statistical analysis of peak pressure differences and ratios was performed in JMP 8.0 (SAS Institute, Cary, North Carolina). The statistical model consisted of an analysis of variance, with the factors graft height (intact, flush, elevated 0.5 mm, elevated 1.0 mm, recessed 0.5 mm, recessed 1.0 mm) and load (200 N, 400 N, 600 N, 800 N). Separate analyses were performed for each combination of lesion location (medial vs lateral) and measurement location (grafted vs opposite facet). The level of significance was chosen as P \ .05. Post hoc comparisons of mean peak pressure differences were made using the Student t test. An overall estimate of the trends in peak pressure ratios was calculated by pooling the data across the 4 loads.
Results
Comparison of Intact and Flush Graft Conditions
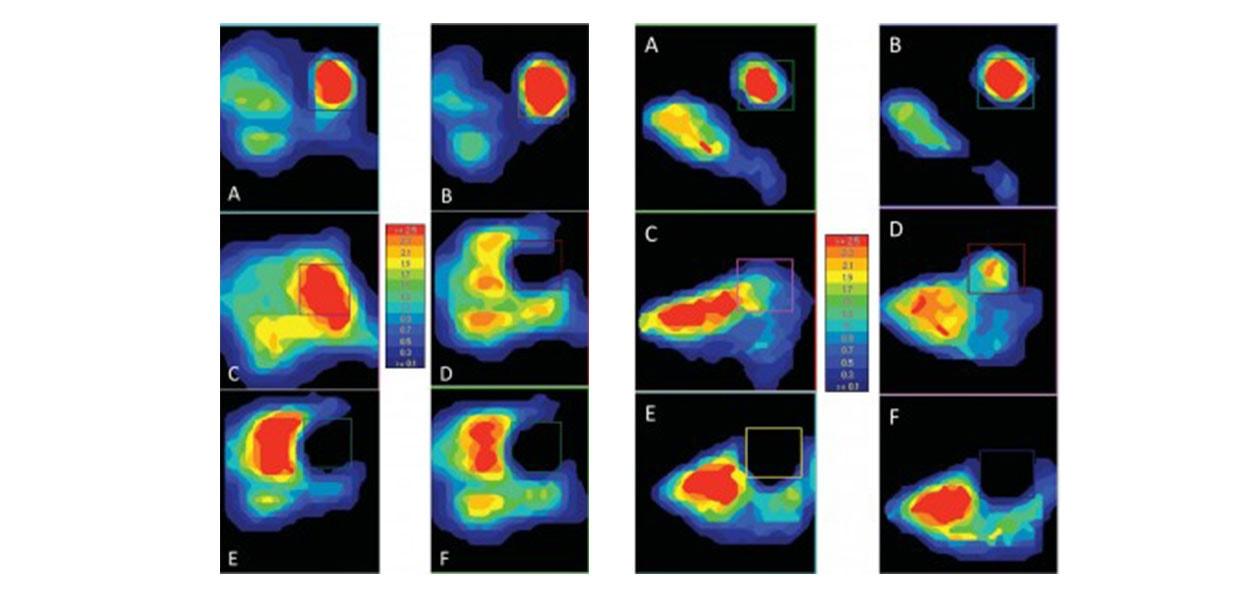
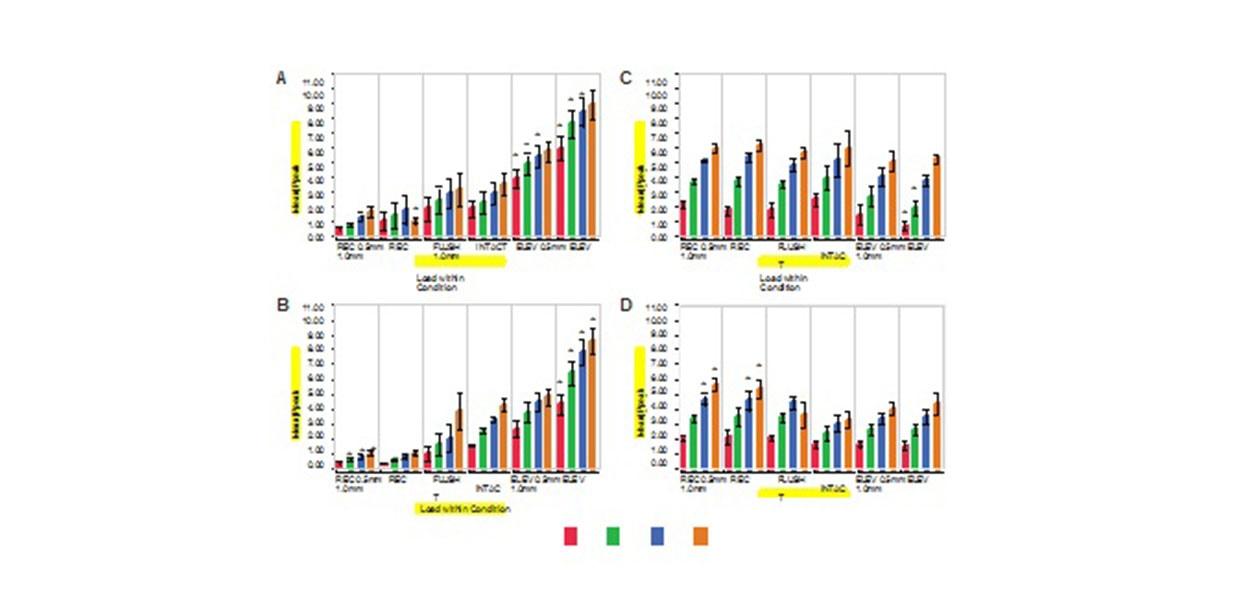
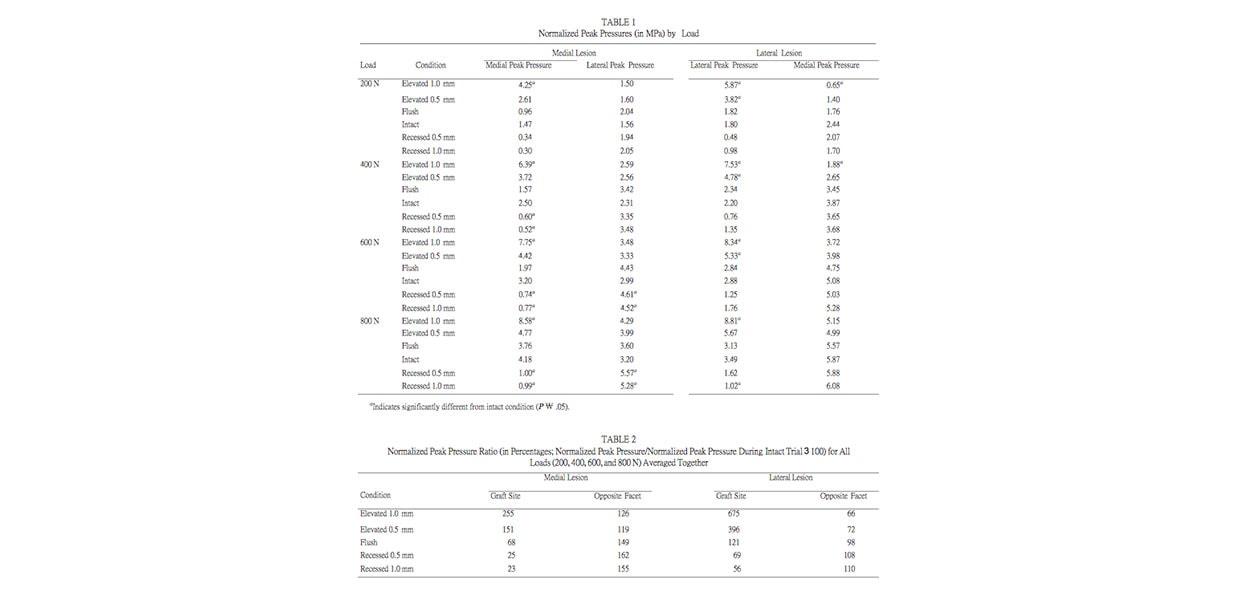
The normalized peak pressure measured at the graft site for the flush condition was not found to be significantly dif- ferent from the intact condition for either medial lesions (Figure 6A) or lateral lesions (Figure 6B). This finding was constant across applied loads (Table 1). In contrast, the normalized peak pressure on the opposite facet of the talar dome was significantly increased during the flush condition (Figures 6C and 6D and Table 2), for the medial (149%) but not the lateral (98%) grafts.
Comparison Across Graft Heights
Normalized peak pressure at the graft site was found to be significantly greater in the elevated 1.0-mm condition than in the flush or intact conditions for both medial and lateral lesions at all loads (Table 1). The increase in peak pressure was particularly evident in the lateral graft conditions, with a pressure ratio of 675% compared with 255% for the medial graft conditions (Table 2). The normalized peak pressure in the 0.5-mm elevated condition was greater than the flush or intact conditions for both medial and lateral lesions, but this difference was not significantat any load (Table 1). However, pooling of the pressures obtained under all 4 loads resulted in pressure ratios of 151% for medial and 396% for lateral lesions. Normalized peak pressure at the graft site decreased significantly in both the recessed 0.5-mm and recessed 1.0-mm conditions for the medial graft conditions (25% and 23%, respectively) but not for the lateral conditions (69% and 56%).
Pressure on Opposite Facet of the Talus
Normalized peak pressure on the opposite facet of the talus increased significantly during the recessed conditions for the medial lesions only at the higher loads (Table 1). The corresponding pressure ratios (Table 2) showed significant increases during both of the recessed conditions for the medial lesions (162% for 0.5-mm recessed and 155% for recessed 1.0 mm).
In contrast, the effect on the opposite side of the talus was reversed for the lateral lesions, such that the normal- ized peak pressure on the opposite facet of the talus decreased significantly during the elevated conditions with the lower loads (Table 1). Similarly, the pressure ratios (Table 2) showed significant decreases during both the elevated conditions for the lateral lesions (72% for ele- vated 0.5 mm and 66% for elevated 1.0 mm).
Discussion
The goal of this study was to quantify changes in joint con- tact pressure with OAT graft height mismatch both at the osteochondral graft and elsewhere in the ankle joint. To our knowledge, this is the first study to quantify this effect in the human talus. We found, as expected, that contact pressure on the graft increased significantly as it was ele- vated above the joint surface. We also found that pressure elsewhere in the joint changed in a reciprocal fashion, such that a recessed graft experiences less pressure while other locations in the joint are subjected to increased pressure.
Effect of Graft Height
Grafts that were elevated relative to the surrounding car- tilage experienced increased peak contact pressures regardless of graft location or applied load. This finding mirrors that of previous studies performed in porcine knees.6,7 On the other hand, when grafts were recessed, we found that peak contact pressures in the region of the graft decreased. This is in sharp contrast to the findings of a previous study,7 which found that recessed grafts experi- enced greater pressures than the intact cartilage. This differ- ence may be attributable to the lower (80-N) loads or the smaller lesions (4.5 mm) that were used in their study. Another possibility is that the pressure-sensitive film that was used in their study is more responsive to high pressures over very small areas, whereas such pressures are averaged over the neighboring sensing elements when the Tekscan sensors are used. A final possibility is that the pressure-sen- sitive film used in their study was recording shear forces, for which it is known to be highly sensitive, and would confound their measurements of normal force.2,14
Joint Pressure Distribution
In the current study during the intact condition, the load was noted to be concentrated on the medial side of the joint. This is in contrast to the findings of 2 other stud- ies8,13 that found higher pressures on the lateral facet dur- ing simulated stance phase. Suckel et al13 examined joint contact pressure in cadaver ankles with simulated gait. Their cadaver model employed a ground-reaction force of
350 N and loaded the extrinsic tendons of the foot in a physiologic way. They found peak contact pressures of 4.7 MPa on the lateral talus and 3.9 MPa on the medial talus during push-off. However, they noted that this load- ing pattern was not uniform and only occurred in 4 of their 8 specimens. When we statically applied 400 N of load, we found average medial and lateral peak pressures to be 2.3 MPa laterally and 3.2 MPa medially. In another study, Michelson et al8 compared static and dynamic loading of the ankle joint with 220 N and found that the pressures at neutral flexion/extension were greater on the lateral talus than on the medial talus (35 vs 20 [units not given]). They also found there was no difference in joint contact pressures between those recorded during static and dynamic loading conditions. The lack of active muscle con- traction in the present study is likely a factor in producing the lower peak pressures and may explain the difference in joint loading pattern between our study and the other 2.
Medial Versus Lateral Lesions
When lateral lesions were examined, the graft had little effect on the overall pattern of joint loading unless it was elevated, in which case it offloaded the medial side. The sit- uation for medial lesions was just the opposite, as the medial side was always loaded unless the graft was recessed, in which case the load shifted to the lateral side. These findings are likely the result of the load distri- bution, which, as discussed above, was found to be concen- trated on the medial side. The clinical importance of this finding is that one must be especially careful to avoid ele- vated grafts on the lateral side as they have the potential to increase pressure on not only the graft, but also on the already diseased lateral facet of the talus.
Comparison With Other Studies
There are a number of important differences in both tech- nique and results between the current study and the preced- ing study performed by Koh et al.7 Their study, in addition to being performed in an animal model, used an 80-N single load, whereas the current study utilized loads of 200, 400, 600, and 800 N applied sequentially. These loads are more physiologic as they roughly correspond to 20 to 80 kg of mass. Koh et al7 looked only at the region surrounding the defect and concluded that peak contact pressures increase both with elevated and recessed graft placements. The cur- rent study examined the pressure across the entire joint, and found that there is an increase with elevated grafts and a decrease with recessed grafts, with reciprocal changes on the contralateral side. This reciprocal loading
has not been previously shown.
Clinical Significance
The findings of this study have a number of important clin- ical implications. The flush placement of the graft is capa- ble of reproducing the joint loading pattern experienced in the intact conditions. If the graft cannot be placed entirely flush because of the shape of its chondral surface, it should be partially recessed, as this will effect a partial unloading of the graft site, giving it a chance to heal. Finally, this study confirms previous findings that elevated grafts are to be avoided as they experience increased pressures, which can lead to graft failure.
Limitations of the Study
There are a number of limitations to this study. First, we did not attempt to replicate physiologic joint loading. We merely recorded contact pressures during the application of static axial loads. There is almost certainly a difference in the joint pressure distribution between these different loading situa- tions, but this difference is unknown. Second, there is a potential for both remodeling and subsidence after the placement of osteochondral grafts in vivo that cannot be rep- licated in vitro; our model can only make predictions about what happens at the time of implantation. Remodeling and subsidence of the graft require further study in either an ani- mal or human model. Finally, we created an idealized defect and filled it with a perfectly matched graft. This eliminated the variability inherent in matching graft size and shape to the defect. However, this required shimming both under and around the graft with nonosseous material. The plastic sleeves that were used to shim around the graft have coeffi- cients of friction and compressibilities that are less than that of bone. It is not known how this may have affected the abil- ity of the graft to settle under load. Moreover, the aluminum shims used to elevate the graft were less compressible than the surrounding cancellous bone of the talus. However, the total thickness of the stacked shims needed was only 2 to 3 mm for the most elevated graft conditions, compared with approximately 10 mm of graft above and 10 mm of talus below. Thus, they likely had a minimal effect on the overall compressibility of the impacted grafts.
There are also some potential sources of error related to the way in which we obtained the pressure maps using the Tekscan system. (1) We chose not to use registration marks on the talus as we were concerned that they might alter the pressure distribution. By not having markers at the edges of the talus, we were unable to tell whether the sensor covered the entire contact surface of the talus during any given trial, and therefore whether it captured the entire transmitted load. However, the Tekscan knee sensor fit very well in each of the ankle joints used in this study. Both direct visu- alization of the sensor within the joint and the absence of pressure at the edge of the sensing area indicated that, in most cases, the joint contact surface was completely covered by the sensor. (2) Since we did not use registration marks, we determined the location of the ROI by visual inspection of the pressure maps. Thus, we were also unable to know for certain whether the ROI was placed accurately over the graft site during the analysis with the Tekscan software. This was particularly problematic during the intact trials, when there was not an obvious image of the circular graft on the pressure maps. (3) The total force recorded by the sensor often differed slightly from the applied load. To account for this, we scaled all pressure measurements to the applied load, but this assumes that we captured the entire contact area in each trial. (4) Also, although only
mm thick, the interposition of the sensor in the joint likely had an effect on joint loading. Previous studies using a measurement device of similar size showed an impact on pressure distributions ranging from 10% to 26%.15 We con- trolled for this effect by placing the same sensor in the same location during each of the trials. It is, however, possible that the interposition changed not only the pressure magni- tudes, but also the pressure distribution pattern.
Conclusion
Flush graft placement can restore near-normal joint con- tact pressures in the ankle at the time of graft placement Small changes in graft height can lead to significant increases in peak contact pressures. These changes are particularly dramatic when the graft is elevated; this not only overloads the graft, but also the entire diseased hemi- talus. On the other hand, even slightly recessed graft placement leads to lower contact pressure in the region of the graft with a corresponding increase in pressure on the healthy opposite facet of the talus. This offloading of the diseased facet of the talus may have a beneficial effect on graft healing. It is thus recommended that osteochon- dral grafts be placed flush with the surrounding surface when possible and slightly recessed when not possible.
References
- Al-Shaikh RA, Chou LB, Mann JA, Dreeben SM, Prieskorn D. Autol- ogous osteochondral grafting for talar cartilage defects. Foot Ankle Int. 2002;23(5):381-389.
- Calhoun JH, Li F, Ledbetter BR, Viegas SF. A comprehensive study of pressure distribution in the ankle joint with inversion and eversion. Foot Ankle Int. 1994;15(3):125-133.
- Easley ME, Latt LD, Santangelo JR, Merian-Genast M, Nunley JA 2nd. Osteochondral lesions of the talus. J Am Acad Orthop Surg. 2010;18(10):616-630.
- Elias I, Zoga AC, Morrison WB, Besser MP, Schweitzer ME, Raikin SM. Osteochondral lesions of the talus: localization and morphologic data from 424 patients using a novel anatomical grid scheme. Foot Ankle Int. 2007;28(2):154-161.
- Hangody L, Kish G, Modis L, et al. Mosaicplasty for the treatment of osteochondritis dissecans of the talus: two to seven year results in 36 patients. Foot Ankle Int. 2001;22(7):552-558.
- Koh JL, Kowalski A, Lautenschlager E. The effect of angled osteo- chondral grafting on contact pressure: a biomechanical study. Am J Sports Med. 2006;34(1):116-119.
- Koh JL, Wirsing K, Lautenschlager E, Zhang LO. The effect of graft height mismatch on contact pressure following osteochondral graft- ing: a biomechanical study. Am J Sports Med. 2004;32(2):317-320.
- Michelson JD, Checcone M, Kuhn T, Varner K. Intra-articular load distribution in the human ankle joint during motion. Foot Ankle Int. 2001;22(3): 226-233.
- Sammarco GJ, Makwana NK. Treatment of talar osteochondral lesions using local osteochondral graft. Foot Ankle Int. 2002;23(8): 693-698.
- Saxena A, Eakin C. Articular talar injuries in athletes: results of micro- fracture and autogenous bone graft. Am J Sports Med. 2007;35(10): 1680-1687.
- Schachter AK, Chen AL, Reddy PD, Tejwani NC. Osteochondral lesions of the talus. J Am Acad Orthop Surg. 2005;13(3):152-158.
- Scranton PE Jr, Frey CC, Feder KS. Outcome of osteochondral auto- graft transplantation for type-V cystic osteochondral lesions of the talus. J Bone Joint Surg Br, 2006;88(5):614-619.
- Suckel A, Muller O, Wachter N, Kluba T. In vitro measurement of intraarticular pressure in the ankle joint. Knee Surg Sports Traumatol Arthrosc. 2010;18(5):664-668.
- Thordarson DB, Motamed S, Hedman T, Ebramzadeh E, Bakshian S. The effect of fibular malreduction on contact pressures in an ankle frac- ture malunion model. J Bone Joint Surg Am. 1997;79(12):1809-1815.
- Wu JZ, Herzog W, Epstein M. Effects of inserting a pressensor film into articular joints on the actual contact mechanics. J Biomech Eng. 1998;120(5): 655-659.